From the Journal: Understanding Injections and Migraine
Guidance on achieving optimal outcomes from injections for migraine treatment
The American Headache Society is committed to keeping its members up to date on the most innovative and meaningful advancements in the realm of headache medicine. One of many initiatives to support that mission is our publication of Headache: The Journal of Head and Face Pain, published ten times per year, which highlights the latest findings in the realm of headache medicine research. Our “From the Journal” series offers a preview of some of the groundbreaking work detailed in Headache.
One of the most widely shared articles in the May issue of Headache was a study on the findings from “Insights into the Functional Anatomy Behind the PREEMPT Injection Paradigm: Guidance on Achieving Optimal Outcomes.” AHS members can click here to read the full report. In the meantime, review the summary below.
Summary: Knowledge of, and adherence to, the standardized PREEMPT injection paradigm supports optimal efficacy and safety in onabotulinumtoxinA injection treatment
The utility of onabotulinumtoxinA (Botox) injections within the PREEMPT paradigm has been widely accepted for years as a viable chronic migraine treatment, but a new study published earlier this year in the Headache journal provides a critical reexamination of best practices, including the precise muscles and techniques clinicians should prioritize to increase the likelihood of effective pain relief.
The report of the study’s findings, titled “Insights into the Functional Anatomy Behind the PREEMPT Injection Paradigm: Guidance on Achieving Optimal Outcomes” and conducted by Dr. Andrew M. Blumenfeld and his colleagues, was centered on the PREEMPT clinical program, formally known as Phase III Research Evaluating Migraine Prophylaxis Therapy.
Blumenfeld and his co-authors confirmed that insufficient understanding of the anatomy and function of individual patients’ head and neck muscles targeted by PREEMPT-guided onabotulinumtoxinA injections can undermine their effectiveness, and prescribed a more individualized approach that considers each patient’s unique anatomy in future administrations of the treatment.
According to the team’s summary of their findings: “Thorough knowledge of the functional anatomy of the muscles involved in the standardized PREEMPT injection paradigm is critical to achieve the efficacy and safety observed in clinical trials. In addition, it is important to assess a patient’s baseline condition to anticipate the risk for unwanted outcomes that may result from treatment.”
While the study generally reaffirmed the safety and efficacy of onabotulinumtoxinA injections in the PREEMPT paradigm as a tool for reducing head and face pain, it also underscored the importance of accurate target muscle localization, injection angles and depths to achieve optimal outcomes and to minimize adverse events including neck pain, muscular weakness and ptosis.
Enjoy access to the full catalogue of content plus a print subscription included with an American Headache Society Membership. Click here to become a member today.
Various depictions of ptosis, one of the most commonly reported injection-related adverse events. (a) No ptosis. (b) Lid ptosis with compensatory frontal overactivity. (c) Pseudo ptosis. (d) Brow ptosis. (e) Medial brow ptosis with compensatory lateral brow elevation.
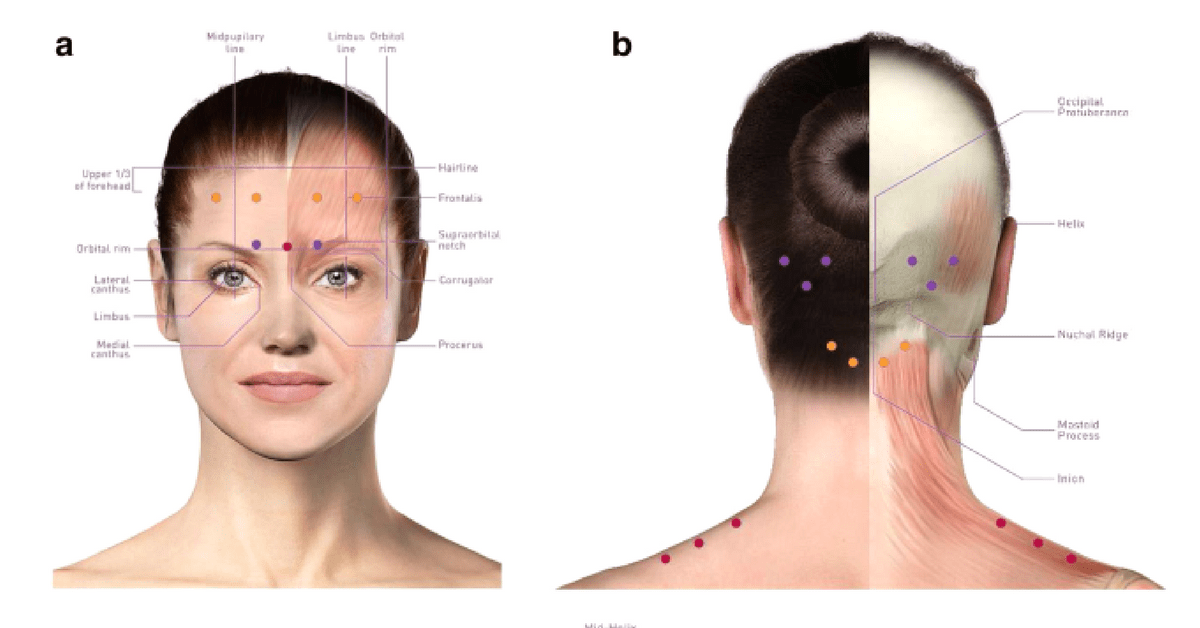
Fixed-site, fixed-dose PREEMPT injection site locations. (a) Corrugator, as depicted by purple dots; procerus, as depicted by the red dot; frontalis, as depicted by orange dots. (b) Occipitalis area, as depicted by purple dots; cervical paraspinal area, as depicted aby orange dots; trapezius, as depicted by red dots. (c) Temporalis, as depicted by purple dots.


